Commercial Pharma Packaging
Commercial pharmaceutical packaging is crucial for ensuring the safety, efficacy, and proper usage of medications. It protects products from external factors that could degrade their quality, complies with regulatory requirements, and enhances patient safety and health outcomes.
Key Functions of Pharmaceutical Packaging:
Protection of Medications
Packaging serves as a barrier against environmental factors such as moisture, light, oxygen, and contaminants, ensuring that medications remain stable and effective until they reach patients.
Safety Features
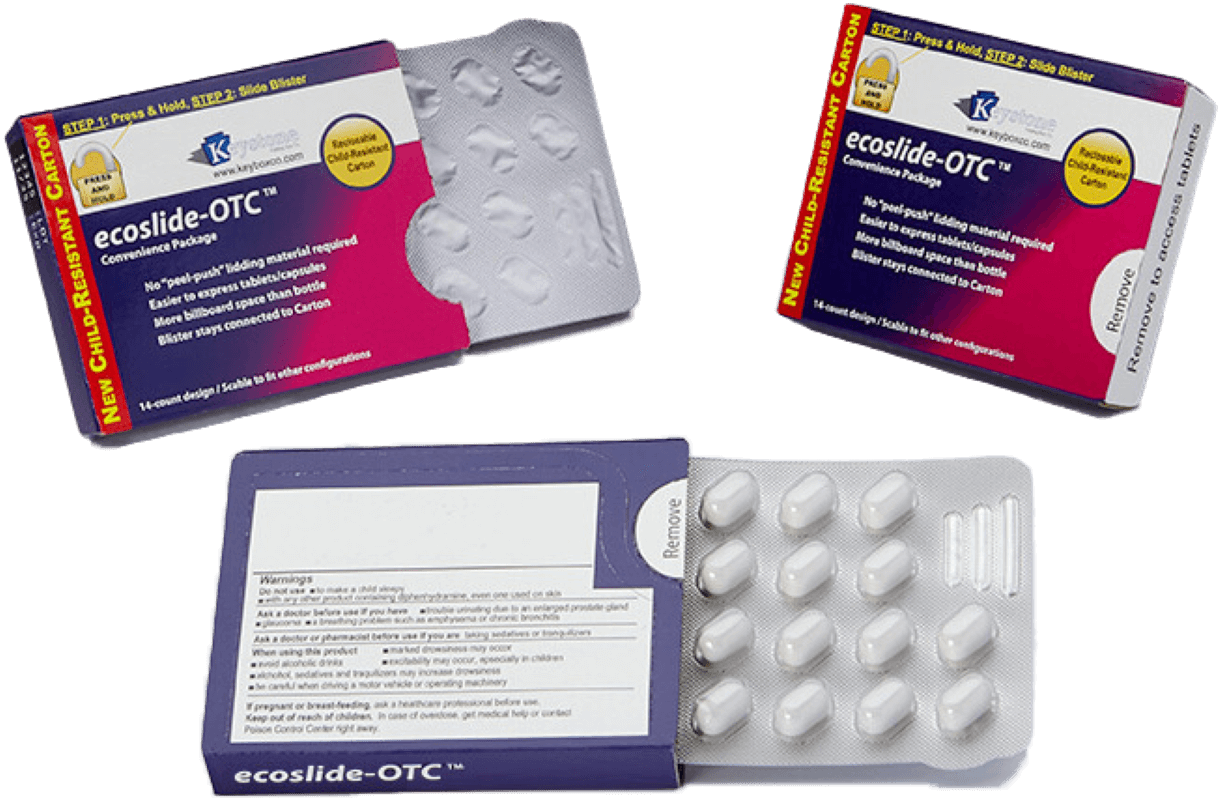
Effective packaging protects patients from harm. It includes child-resistant closures to prevent accidental ingestion, tamper-evident seals to indicate compromised packages, and clear labeling to convey vital information about dosage, usage instructions, and potential side effects.
Regulatory Compliance
Pharmaceutical packaging must adhere to stringent regulations set by health authorities. This includes using safe materials and providing accurate labeling that meets legal standards.
Sustainability Initiatives
Utilizing responsibly sourced packaging materials that are recyclable or compostable enables pharmaceutical companies to positively impact environmental conservation.
Convenience and Usability
User-friendly packaging design ensures that patients can safely and correctly access their medications. Features may include easy-open packaging and clear instructions.
Brand Identity
Packaging plays a significant role in branding and marketing, helping differentiate products through logos, colors, and designs that enhance product recognition and consumer trust.